Hydrogen Storage in Nanocavities
Abstract
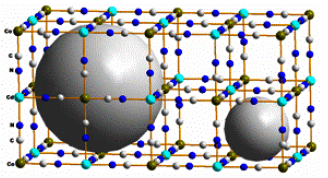
Hydrogen, because of its large combustion heat, 572 kJ/mol, three times that of gasolines, and an environmentally compatible byproduct, water, is being considered as an alternative to fossil fuels derivatives; particularly in vehicular applications. The application of hydrogen as combustible in mobile technologies involves three challenges: the production from the water splitting, for instance; the availability of a reversible, safe and economically viable storage method; and its use through appropriate fuel cells where the chemical energy is directly transformed in electricity. Nature practically has no free H2. From this fact, H2 must be produced. It is a secondary energy bearer, not a primary source like petroleum. The storage is probably the main of these challenges because H2 has very low critical temperature (Tc = 32.97 K) and at ordinary temperatures it is supercritical gas. The H2 liquefaction is possible when the strength for the H2-H2 interactions (of van der Waals and quadrupole-quadrupole type) surpasses the thermal energy (kT). This suggests the possibility of be able the H2 storage at temperatures above Tc through a strong H2-surface interaction in solids of extended surface, layered or nanoporous materials, for instance. In this contribution the possible H2- surface interactions that could be present in such storage method and their relative contributions to the H2 adsorption energy in materials of extended surface are discussed. The role of these interactions is dicussed from experimental results on H2 in nanocavities. The state of art and perspectives on this storage method are also evaluated.
This work is licensed under the Creative Commons Attribution-NonCommercial 4.0 International (CC BY-NC 4.0) license.








