Structural, EPR and Optical Studies on Cu-doped ZnO Nanoparticles Synthesized by the Sol-Gel Method at Different Calcination Temperatures
Abstract
In this work, pure ZnO and Cu-doped ZnO nanoparticles (Zn1-xCuxO, x = 3 at.%) were synthesized using the sol-gel method at three different calcination temperatures (TC = 773 K, 823 K and 873 K.). The particles were analyzed by atomic absorption spectroscopy (AAS), X-ray diffraction (XRD), electron paramagnetic resonance (EPR) at different measurement temperatures and diffuse reflection spectroscopy (DRS). All samples have wurtzite structure and the formation of a CuO was observed as additional phase. The Cu-doped nanoparticles have smaller size than the
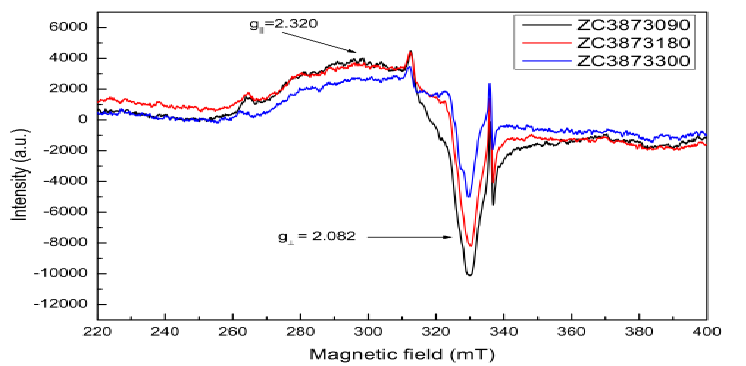
pure ZnO particles. EPR analysis shows that in samples of ZnO there is presence of superficial defects (g = 2.036, g = 1.967 and g = 1.958) in increased amount than in doped samples. Both samples shows Zn vacancy with g = 2.004. The Cu EPR signal was simulated by an anisotropic Spin-Hamiltonian with the values of gt = 2.082 and gp = 2.320 and indicating that the Cu2+ ions in the sample have a local configuration with axial symmetry. Cu doped samples reveal a smaller band gap for the Cu-containing samples than for pure ZnO samples. Additionally, the gap of the Cu-containing samples decreases with increasing calcination temperature. Samples calcinated at 823 K are anti-ferromagnetic or ferrimagnetic at temperature lower than TN = 139 K while samples calcinated at 873 K suffer a transition from paramagnetism to ferromagnetism which is characterized by a Curie temperature of
OC = 137 K.
This work is licensed under the Creative Commons Attribution-NonCommercial 4.0 International (CC BY-NC 4.0) license.








